Research teams
Molecular bases of human diseases
Molecular mechanisms in women’s health and disease

Ovaries produce and release mature oocytes and act as an endocrine organ by secreting sex hormone. The ovarian surface epithelium (OSE) repairs itself after ovulation but is also susceptible to genetic and epigenetic changes over time, potentially increasing cancer risk. Stem cells in the OSE contribute to tissue repair, and recent research explores how inflammation marks long-term with an “epigenetic memories” that affect cell identity and cancer development. Endocrine factors like androgens and insulin also influence ovarian health, with hyperandrogenism linked to Polycystic Ovarian Syndrome (PCOS) and ovarian cancer.
The regulation of ovaries and reproductive tract is complex and coordinated by hormones produced by the endocrine system such as the hypothalamus-pituitary-ovarian axis.
The thymus, well known as the site of immune T cell development, is also an endocrine gland. It controls female fertility via thymic hormones such as thymulin. The absence of thymus reduces reproductive capacity in animals, indicating that this axis plays an essential role. Hormonal dysregulation may disrupt this communication by impairing the function of each individual organ.
Our experimental approaches integrate multiple disciplines – including cell biology, genomics, and physiology – by combining in vivo and in vitro models, and leveraging advanced imaging, sequencing, and spatial technologies.
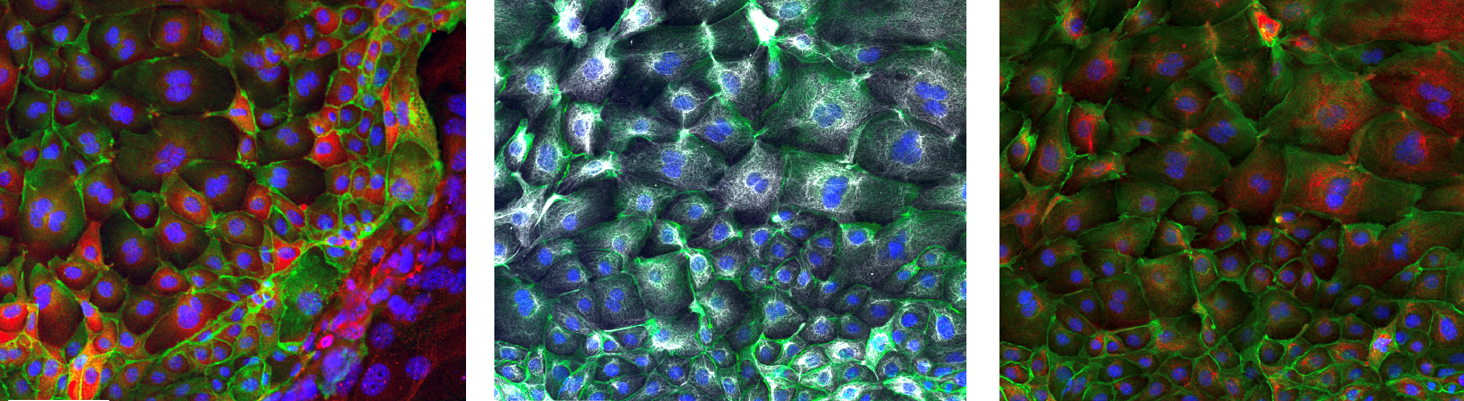
OSE undergoes several physiological insults (ovulations) during the lifespan of female individuals. We are testing how recurrent ovulation ruptures alter the OSE epigenome and increase the chances of developing pre-malignant transformations later in life.
We perform a thorough molecular characterisation of the OSE before and upon ovulations throughout the entire lifespan of female mice thanks to existing reporter line. In parallel, we characterise human OSE obtained from different cohorts of patients in collaboration with University hospital CHU in Montpellier and Institut Paoli Calmettes in Marseille.
2- What is the impact of hormonal imbalance on ovarian epithelium?
Endocrine imbalance such as exposure to high doses of androgens in PCOS disease might alter the ovarian cell components. Ovarian epithelium responds to environmental changes by upregulating the corresponding receptors, such as androgens receptor, with a potential impact on OSE homeostasis during female lifespan.
We establish a PCOS mouse model (Prenatal Anti Mullerian Hormone (PAMH) treatment during pregnancy) most akin to the human condition to explore how the OSE responds to different endocrine inputs from early development to ageing. We will take advantage of in vitro primary cultures of ovarian epithelial cells to dissect the molecular mechanisms triggered by PCOS hormonal levels.
3- What are the mechanisms at the base of thymus-ovarian axes?
Based on the long-known but poorly explored fact that the thymus regulates female fertility and ovarian function, we seek to understand how this is regulated.
Using thymus-deficient mice, two paths will be studied: thymic influence on pituitary hormone control and direct thymic regulation of ovarian cells.
In addition, we are also interested to underpin how ovarian hormonal imbalance affect thymus development and function (e.g. in PCOS murine model).


- AK Reuschl, LG Thorne, MVX Whelan, R Ragazzini , W Furnon, VM Cowton, G De Lorenzo , D Mesner, JLE Turner, G Dowgier, N Bogoda, P Bonfanti, M Palmarini, AH Patel, C Jolly , GJ Towers Evolution of enhanced innate immune suppression by SARS-CoV-2 Omicron subvariants. Nat Microbiol. 2024 Feb;9(2):451-463.
- Roberta Ragazzini, Stefan Boeing, Luca Zanieri, Sara Watson, Antoniana Batsivari, Kerol Bartolovic, Ana Agua-Doce, Adrian C. Hayday, Dominique Bonnet and Paola Bonfanti Defining the identity and multilineage potency of a unique epithelial stem cell in the human postnatal thymus. Developmental Cell 58, 1–19.
- Dejan Mesner, Ann-Kathrin Reuschl, Matthew V. X. Whelan, Taylor Bronzovich, Tafhima Haider, Lucy G. Thorne, Roberta Ragazzini, Paola Bonfanti, Greg J. Towers, and Clare Jolly SARS-CoV-2 evolution influences GBP and IFITM sensitivity. PNAS Jan 2023; DOI: 10.1073/pnas.2212577120.
- Sara A. Watson, Yousef Javanmardi, Luca Zanieri, Somayeh Shahreza, Roberta Ragazzini, Paola Bonfanti, Emad Moeendarbary Integrated role of human thymic stromal cells in hematopoietic stem cell extravasation. Bioengineering&Translational Medecine, Nov 2022; DOI: 10.1002/btm2.10454
- Lucy G. Thorne, Mehdi Bouhaddou, Ann-Kathrin Reuschl, Lorena Zuliani-Alvarez, Ben Polacco, Adrian Pelin, Jyoti Batra, Matthew V. X. Whelan, Myra Hosmillo, Andrea Fossati, Roberta Ragazzini, Irwin Jungreis, Manisha Ummadi, Ajda Rojc, Jane Turner, Marie L. Bischof, Kirsten Obernier, Hannes Braberg, Margaret Soucheray, Alicia Richards, Kuei-Ho Chen, Bhavya Harjai, Danish Memon, Joseph Hiatt, Romel Rosales, Briana L. McGovern, Aminu Jahun, Jacqueline M. Fabius, Kris White, Ian G. Goodfellow, Yasu Takeuchi, Paola Bonfanti, Kevan Shokat, Natalia Jura, Klim Verba, Mahdad Noursadeghi, Pedro Beltrao, Manolis Kellis, Danielle L. Swaney, Adolfo García-Sastre, Clare Jolly, Greg J. Towers, Nevan J. Krogan. Evolution of enhanced innate immune evasion by SARS-CoV-2. Nature, 2021; DOI: 10.1038/s41586-021-04352-y
- Sara Campinoti*, Asllan Gjinovci*, Roberta Ragazzini*, Luca Zanieri, Linda Ariza- McNaughton, Marco Catucci, Stefan Boeing, Jong-Eun Park, John C. Hutchinson, Miguel Munoz-Ruiz, Demetra E. Phylactopoulos, Constance Maurer, Pierluigi G. Manti, Gianluca Vozza, Giuseppe Testa, Hans J. Stauss, Sarah A. Teichmann, Neil J. Sebire, Adrian C. Hayday, Dominique Bonnet, and Paola Bonfanti Reconstitution of a functional human thymus by clonogenic stromal stem/progenitor cells and natural whole organ scaffolds. Nature Commun, 2020 Dec 11;11(1):6372. doi: 10.1038/s41467-020-20082-7
- Jong-Eun Park, Rachel A. Botting, Cecilia Domínguez Conde, Dorin-Mirel Popescu, Marieke Lavaert, Daniel J. Kunz, Emily Stephenson, Roberta Ragazzini, Elizabeth Tuck, Anna Wilbrey-Clark, John Ferdinand, Simone Webb, Daniel Maunder, Niels Vandamme, Krishnaa Mahbubani, Krzysztof Polanski, Lira Mamanova, Andrew Fuller, Andrew Filby, Gary Reynolds, David Dixon, Kourosh Saeb-Parsy, Steven Lisgo, Deborah Henderson, Roser Vento-Tormo, Kerstin Meyer, Yvan Saeys, Paola Bonfanti, Menna Clatworthy, Tom Taghon, Muzlifah Haniffa,, Sarah A. Teichmann. An atlasof human thymic development reveals single cell dynamics of TCR repertoire formation Science. 2020, Feb 21;367(6480).




