Research teams
Genetics, cell biology and development department
Development of pathology of the gonad

In mammals, the chromosomal sex (XX or XY) is established at fertilization. However, the undifferentiated embryonic gonad, or genital crest, remains bipotential until the stage of gonadal determination, at which point it differentiates into either an ovary or a testis.
This event occurs at approximately eleven days of embryonic life in mice and at approximately five weeks of gestation in humans.
The choice between the testicular and ovarian pathways is made in undifferentiated precursors of the genital crest, which differentiate into supporting cells: the testicular Sertoli cells or the ovarian pre-granulosa cells.
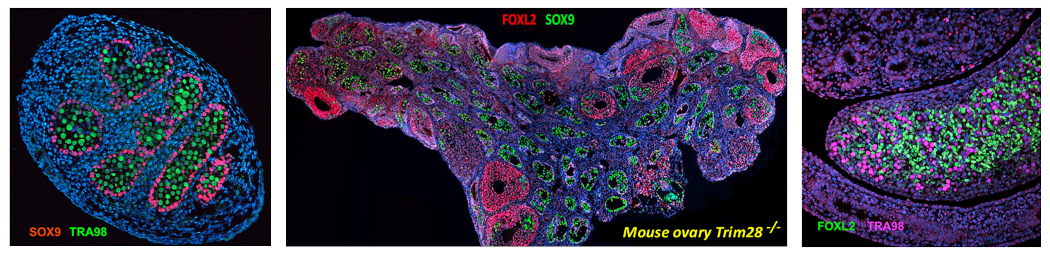
In XY gonads, the expression of SRY, which is carried by the Y chromosome, activates the SOX9 gene, which controls Sertoli cell differentiation.
In XX gonads, the -KTS splice variant of WT1 associated with the WNT4/RSPO1 pathway leads to expression of the transcription factor FOXL2, which controls pre-granulosa cell differentiation.
Following their determination, the supporting cells orchestrate the development of the gonads, resulting in functional organs that produce sex hormones and gametes after puberty. Any disruption to these processes can result in disorders of sexual development, often associated with infertility, which can in turn lead to complete sex reversal.
Furthermore, supporting cells play a crucial role in maintaining the sexual identity of adult gonads. Our recent research has revealed that targeted deletion of the Trim28 gene in ovarian somatic cells induces their post-natal reprogramming into testicular cells. The process entails the trans-differentiation of granulosa cells into Sertoli cells, which ultimately leads to postnatal gonadal sex reversal.
In addition to studying testicular and ovarian determination processes, our group uses murine genetics to elucidate the mechanisms of differentiation and maintenance of the adult sexual phenotype. To this end, we use histological, transcriptomic, genomic and biochemical approaches to study our mouse models.
Deciphering the role of the heterochromatin mark H3K9me3 in mammalian testicular determination
In mammals, the embryonic bipotential gonad can develop either as a testis or an ovary in XY and XX embryos respectively and depends on antagonistic pro-testis and pro-ovarian pathways. Testis development is initiated by the expression of the SRY gene carried by the Y chromosome, that in turn induces the expression of the master transcription factor SOX9 leading to the differentiation of Sertoli cells. These supporting cells produce crucial factors that further orchestrate testis morphogenesis and differentiation. If the “pro-testicular” effects of SRY and SOX9 transcription factors are well documented, little is known about how the ovarian program is repressed during testis development. This is an important issue as the abnormal activation of the female program in XY individuals can lead to sex reversal, one type of disorders of sexual development (DSD). A genetic diagnosis is achieved in only 40% of human DSD patients analyzed by exome sequencing, indicating that additional levels of regulation control gonadal development.
We have recently discovered that sex-determination induces a strong sexual dimorphism for the tri-methylation on the lysine 9 of the histone H3, a marker of heterochromatin regions in mice. While being maintained in testis supporting cells, the repressive mark H3K9me3 is strongly reduced in their female counterpart, the granulosa cells. We hypothesize that an unexpected level of genomic regulation involving heterochromatin and H3K9me3 deposition contributes to sex-determination through the lock-down of the female genes in the testis. Moreover, as H3K9me3 is an important component of DNA repeats, a sexual dimorphism might exist at the level of the repetitive fraction of the genome composing crucial chromosomal structures such as the telomeric and pericentromeric regions.
For the present project we have defined several objectives that propose to investigate:
- The role of heterochromatin on sex determination through the study of multiple sex-reversed mutants and the role of the histone methylase SETDB1 as a central actor of male-specific heterochromatin.
- Using genomic profiling by the CUT&RUN technique on purified Sertoli or granulosa cells, we will test a model where SOX9 would induce the redeployment of SETDB1 and thus of H3K9me3 on chromatin via their common partner TRIM28. In addition, we will analyse the histone marks associated with transcriptional activation or repression.
This project will unravel a new level of regulation of gonadal sex-determination. It will determine whether male-specific heterochromatin formation is a cause or a consequence of sex determination. Moreover, it will provide new perspectives for the analysis of unresolved DSD patients in human. Insights we gain from this project might also help explaining the differences of some disease incidences between man and woman.
Roles of Trim28, a Sox9 partner, in sexual differentiation
Using a genetic approach in mice, we are currently investigating the role of Trim28 in embryonic and adult supporting cell lineages.
Since complete knockout is lethal in early embryonic stages, we have used a conditional “floxed” allele associated with Sf1-Cre or Wt1-CreERT2 to inactivate Trim28 in granulosa and Sertoli cells at different stages of development and after birth.
Transcriptomic, ChIP-seq, and in vitro approaches are also being used to decipher the role of Trim28 in maintaining ovarian identity and in sexual differentiation.


















- Stévant, I., Gonen, N. and Poulat, F. (2024) Transposable elements acquire time- and sex-specific transcriptional and epigenetic signatures along mouse fetal gonad development. Frontiers in Cell and Developmental Biology, 11, 1327410.
- Boizet-Bonhoure, B., Déjardin, S., Girard, M., Durix, Q., Poulat, F. and Philibert, P. (2024) Adenomyotic Lesions Are Induced in the Mouse Uterus after Exposure to NSAID and EE2 Mixtures at Environmental Doses. International Journal of Molecular Sciences, 25.
- Philibert, P., Stévant, I., Déjardin, S., Girard, M., Sellem, E., Durix, Q., Messager, A., Gonzalez, A.-A., Mialhe, X., Pruvost, A., Poulat, F. and Boizet-Bonhoure, B. (2023) Intergenerational effects on fertility in male and female mice after chronic exposure to environmental doses of NSAIDs and 17α-ethinylestradiol mixtures. Food and Chemical Toxicology, 182, 114085.
- Philibert, P., Dejardin, S., Girard, M., Durix, Q., Gonzalez, A.A., Mialhe, X., Tardat, M., Poulat, F. and Boizet-Bonhoure, B. (2023) Cocktails of NSAIDs and 17alpha Ethinylestradiol at Environmentally Relevant Doses in Drinking Water Alter Puberty Onset in Mice Intergenerationally. Int J Mol Sci, 24.
- Rossitto, M., Dejardin, S., Rands, C.M., Le Gras, S., Migale, R., Rafiee, M.R., Neirijnck, Y., Pruvost, A., Nguyen, A.L., Bossis, G., Cammas, F., Le Gallic, L., Wilhelm, D., Lovell-Badge, R., Boizet-Bonhoure, B., Nef, S. and Poulat, F. (2022) TRIM28-dependent SUMOylation protects the adult ovary from activation of the testicular pathway. Nat Commun, 13, 4412.
- Philibert, P., Dejardin, S., Pirot, N., Pruvost, A., Nguyen, A.L., Bernex, F., Poulat, F. and Boizet-Bonhoure, B. (2021) In the mouse, prostaglandin D2 signalling protects the endometrium against adenomyosis. Mol Hum Reprod, 27.
- Rossitto, M., Ollivier, M., Déjardin, S., Pruvost, A., Brun, C., Marchive, C., Nguyen, A.L., Ghettas, A., Keime, C., de Massy, B., Poulat, F., Philibert, P. and Boizet-Bonhoure, B. (2019) In utero exposure to acetaminophen and Self-Assessment Document for Research Units 2025-2026 Evaluation campaign – Group A Research Evaluation Department 173 ibuprofen leads to intergenerational accelerated reproductive aging in female mice. Communications Biology, 2, 310.
- Rossitto, M., Marchive, C., Pruvost, A., Sellem, E., Ghettas, A., Badiou, S., Sutra, T., Poulat, F., Philibert, P. and Boizet-Bonhoure, B. (2019) Intergenerational effects on mouse sperm quality after in utero exposure to acetaminophen and ibuprofen. FASEB J, 33, 339-357.




